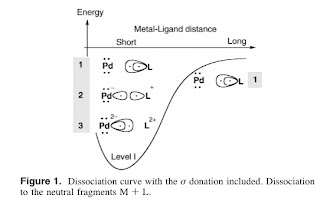
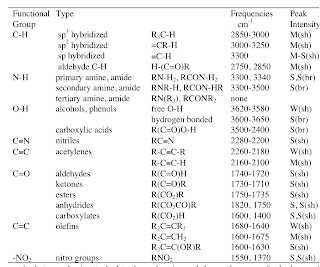
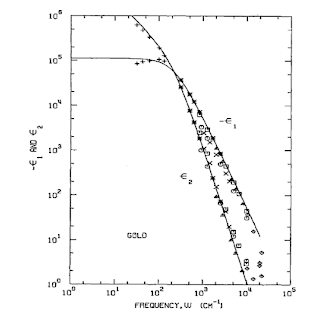
Breaking down the physics-chemistry divide

Chemists and physicists tend to talk different languages, including when discussing the same thing. One important parallel is the common concept of the molecular orbitals in a molecule and the energy bands in a crystal. Specifically, the Huckel method to describe electronic properties of conjugated organic molecules is identical to the tight-binding method in solid state physics. Yet these important parallels seem to rarely be pointed out in textbooks. [One exception is a brief mention in Walter Harrison's Electronic Structure and the Properties of Solids ]. Recently when I have taught solid state I have pointed out the connection and sometimes worked through the nice treatment of Huckel theory in chapter 8 of the classic book Coulson's Valence by Roy McWeeny. Besides showing the molecule-solid connection this can illustrate a few useful things including: How Bloch's theorem works in a finite system. How energy bands emerge in the thermodynamic limit (see above).